Abstract
Introduction
Multiple studies in patients with B-cell lymphomas (BCL) have shown that TP53 is associated with inferior therapeutic efficacy and survival outcomes. Current treatment algorithms for chronic lymphocytic leukemia (CLL) and mantle cell lymphoma (MCL) are tailored for patients harboring a TP53 mutation using a different therapeutic strategy. Regardless, targeted next generation sequencing (tNGS) is not standard practice in the diagnostic workup of lymphomas. In solid malignancies, e.g., ovarian cancer (PMID: 35752743), our institute has shown that p53 immunohistochemistry (IHC) is a good and widely used marker to assess TP53 mutations. In lymphomas however, this correlation is not yet as clearly established. Here we evaluate the accuracy of p53 IHC as a surrogate marker for TP53 mutational analysis in a large cohort of BCL.
Method
For this retrospective study 278 consecutive patients diagnosed with BCL between 2014-2022 were selected. The cases were diagnosed according to the revised 4th edition WHO classification (2016), and included diffuse large B-cell lymphoma (DLBCL) not otherwise specified (NOS, n=145), primary cutaneous DLBCL leg type (PCDLBCL LT, n=13), high grade B-cell lymphoma (HGBCL, n=8), primary central nervous system lymphoma (PCNSL, n=11), primary mediastinal B-cell lymphoma (PMBCL, n=3), Burkitt lymphoma (n=4), mantle cell lymphoma (MCL, n=18) and a diverse group of indolent mature B-cell neoplasms (MBCN, n=76, specified in figure 1).
P53 IHC staining was performed using the Dako Autostainer Link 48 (Dako Omnis, Glostrup, Denmark), according to standard procedures. For each case, p53 IHC was blindly and independently assigned by one junior (LMH) and one senior (PMJ) hematopathologist to one of the following categories: abnormal (complete absence OR staining > 50% of tumor cells) or wild type (WT) (staining 1-50% of tumor cells). All discordant cases were discussed at a consensus meeting attending both pathologists and assigned a definitive category. The inter-rater reliability was measured with Cohen's kappa coefficient.
Pathogenic variants of TP53 for all cases were identified with an in-house developed tNGS panel (LYMFv1), including 52 B-cell lymphoma-relevant genes and covering all exons of TP53, as described before (PMID: 34478526).
Results
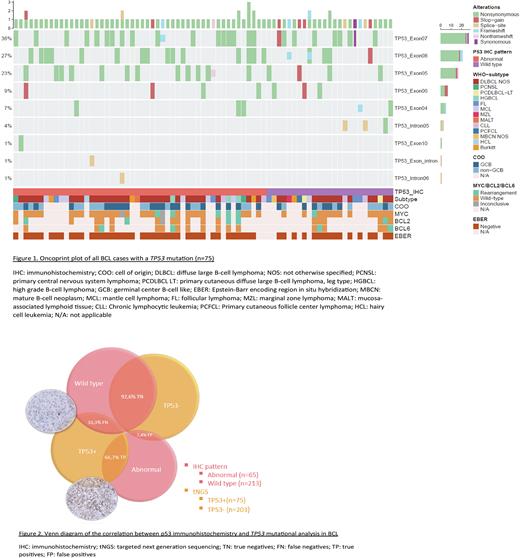
IHC showed 213 p53 WT and 65 abnormal. Kappa correlation of the p53 IHC assessment between the two pathologists was 0,82, indicating a strong level of agreement. tNGS identified a TP53 mutation in 75 patients (26,9%), of which DLBCL NOS was the largest subgroup (n=43), followed by MBCN (n=14) and MCL (n=7). Overall, most TP53 mutations were non-synonymous (80,5%) and localized in exon 7 (36%), as shown in the Oncoprintplot (figure 1). In MBCN the most frequent location was exon 5 (33%), while in DLBCL as the largest subgroup, mainly exon 7 (38%) was affected.
The overall accuracy of p53 IHC was 85,6% (95% CI 80,9-89,5%) for the presence of TP53 mutation. The corresponding sensitivity and specificity were 66,7% (95% CI 56,0-77,4%) and 92,2% (95% CI 88,5-95,9%), respectively.
25 BCLs showed a false-negative (FN) p53 WT IHC, despite detection of TP53 mutation. MBCN was the least accurate performing group (8/14 FN), followed by DLBCL NOS (13/43 FN). On the other hand, there were 15 BCLs with a false-positive (FP) abnormal IHC staining without identification of a TP53 mutation (figure 2). Again, MBCN was the lowest performing group (7/62 FP), but the overall specificity was high.
Conclusion
Our data indicate that the performance of p53 IHC is not sufficient as a surrogate marker for TP53 mutations in the diagnostic workup of lymphoma patients. Although the specificity is high with a low amount of FP, the sensitivity is relatively low with a high number of FN. It seems that misinterpretation of the p53 IHC is not an adequate explanation for the low sensitivity, considering the strong inter-rater reliability and the re-assessment of all discordant cases.
By utilizing only p53 IHC there is a significant chance a TP53 mutation is missed, resulting in misevaluation of a high-risk patient. Therefore, molecular analysis is recommended in all patients with BCL, especially for further development of risk-directed therapies based on TP53 mutation status.
Disclosures
Diepstra:Takeda: Membership on an entity's Board of Directors or advisory committees, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal